
Its electron configuration of 2-8-19-1 allows it to work well as a pure element and in a variety of compounds. Potassium and chromium are the other two. It is located in the fourth row (period) on the table of elements and is one of three elements that has one electron in its outer orbital. This transition element is well known for the two colors that are reddish when pure and a turquoise color when copper is oxidized. The fourth row of the periodic table has transition metals ranging from scandium (21) to zinc (30). 
Remember that the first eight were placed during our trip through the third period/row. For the fourth period/row, all of these electrons build the third shell to a maximum of 18 electrons. chemistry logo science logo atom laboratory logo chemical formula. This element is one of the transition elements that doesn't place the additional electrons in the outer shell, but in the one underneath. Copper element, the concept of chemistry. So remember when you look at our breakdown that the electrons aren't always in a nice neat order as shown here. This table has links to all the properties of copper included within WebElements. They are found in clouds that can have different shapes that include spheres and dumbbell-like shapes. As you learn more about atomic structure, you will learn that the electrons don't stay in defined areas around the nucleus. Copper is a transition metal at the top of group 11 on the periodic table, along with silver and gold. The electrons like to be in separate shells/orbitals. The element copper has symbol Cu and atomic number 29. In an atom, the electrons spin around the center, also called the nucleus. Each of those colored balls is an electron. If you think this is a little over your head, go back and look at the elements 1-18 that have organizations that are a little more simple. Let's take a look at the arrangements of electrons in the basic elements (left and right sides of the table) of period four and the more complex arrangements of the transition elements (in the middle of the row). More about the history and places to find copper. 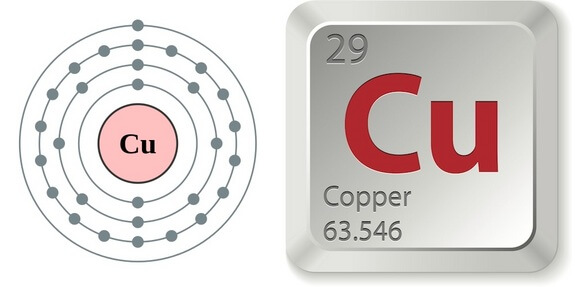
You may have an easy way to know the number of electrons in a neutral atom, but the placement of those electrons gets a little more complex. Find Cu Copper Chemical Element Periodic Table stock images in HD and millions of other royalty-free stock photos, illustrations and vectors in the Shutterstock. It is located in the fourth row (period) on the table of elements and is one of three elements that has one electron in its outer orbital. Now we're working with the fourth period/row in the table of elements. Relative atomic mass: 63.546 (3) Copper has been known since ancient times. It tells you the mass of one atom, how many pieces are inside, and where it should be placed on the periodic table. That box on the left has all of the information you need to know about one element.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
